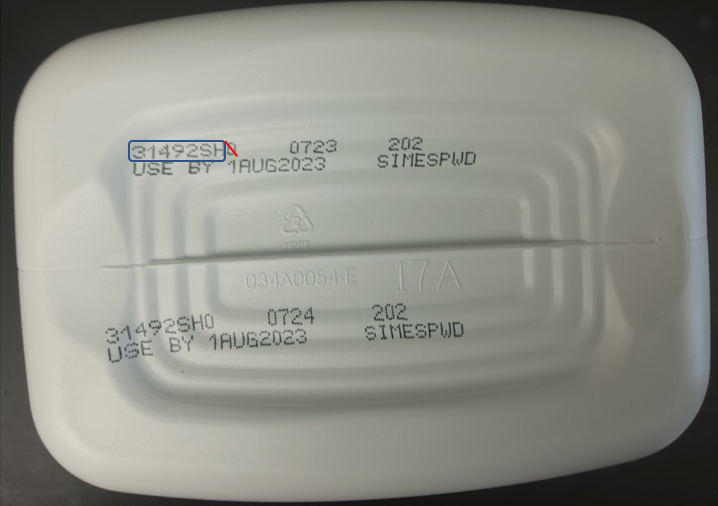
- Impacted lots will start with a 22 through 37 excluding the leading alphanumeric character - Example: L31465SH00
- Only one lot number can be searched at a time
- Locate the lot number on the bottom of your container
- Enter the first 7 characters of the lot number in the lot search field
- If the lot number on your container starts with a letter it represents a lot sold internationally – please exclude the leading letter when entering the lot number
- If the lot number entered in the lot search field starts with a letter you will receive an error message to ensure your entry starts with a number
- If the lot number entered in the lot search field is part of the recall you will be asked to enter additional information
- If the lot number entered in the lot search field is not part of the recall you will receive a message that your lot number entered is not impacted
- If you are not able to enter the lot number and would like to speak with a customer service representative, if in the United States please call 1-800-986-8540 or if international please contact the number on the product label
Examples of containers indicating the characters to enter in the lot search field:

Abbott News
TWITTER
Abbott Careers
FACEBOOK
Abbott Careers
LINKEDIN
Abbott Global
YOUTUBE
Abbott Global
INSTAGRAM