Leadless Pacemaker Keeps the Beat? Bravo, Aveir! Bravo!
Aveir could treat slow heart rhythms — and can be retrieved later — all through minimally-invasive procedures.
Dec. 14, 2021
- Copy Link
- Share on X
- Share on Facebook
- Share on Linkedin
Through the chords of life — whether it's moving adagio (slow and stately) or allegro (quickly) for you — your heart works in a four-part harmony.
Start at the top of the range:
The right atrium, the highest point — like a soprano — of the blood's movement into and through the heart.
Then moving down to the right ventricle, the alto.
Then the blood courses, vivace, through the pulmonary artery to the lungs — where it deepens with oxygenation, the tenor — and back to the left atrium.
From there, down to the left ventricle — the bass — the next contraction a boom of enriched blood heading out of the aorta and to the body, only to return da capo, hitting that high note at the top before starting the run all over again.
But when some part of the heart loses time, a conductor can restore form to its proper pace. We call that maestro a pacemaker.
But what if the maestro did not have his baton to deliver energy to the orchestra?
Would that not be like a pacemaker not having its cardiac leads?
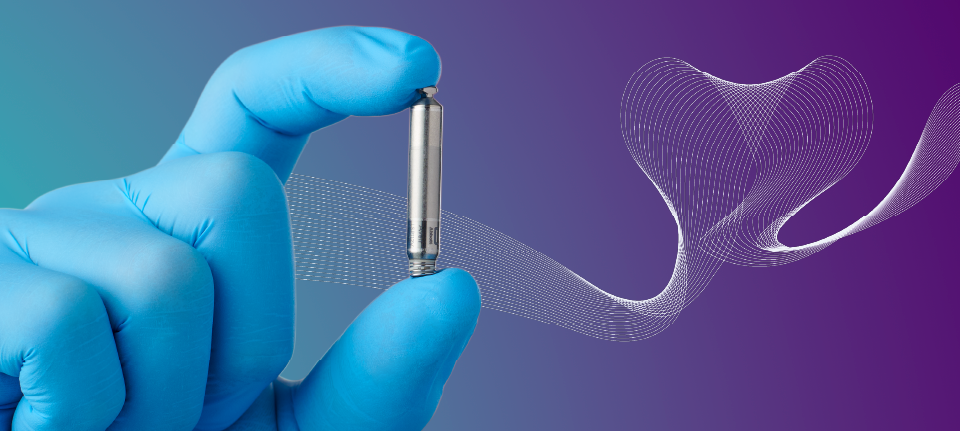
Let us introduce you to Aveir, the leadless pacemaker.
Meet the New Conductor
Late-breaking data from the global Leadless II phase 2 IDE study suggests Aveir VR leadless pacemaker, once approved, could benefit people who require the use of a pacemaker to treat slow — adagio — heart rhythms.
Among the study's findings, Aveir was successfully implanted in 98% of patients and, at six weeks post-implant, 96% of patients had no serious adverse device effects.
Unlike traditional pacemakers, such as our Assurity MRI, leadless pacemakers do not require an incision in the chest to implant the device or insulated wires (cardiac leads) to deliver energy to the heart.
Instead, they are implanted directly into the heart's right ventricle — the alto — via a minimally invasive procedure.
"What makes the Aveir system unique is that it is the world's only leadless pacemaker specifically designed to be retrieved when the device needs to be replaced or if a patient's therapy needs to be changed," said Dr. Vivek Y. Reddy, Helmsley Trust professor of medicine, director of cardiac arrhythmia services, Mount Sinai Hospital.
Reddy also outlined Aveir’s additional potential advantages:
- Long battery life
- Ability to expand to a true dual chamber pacemaker, once software update approved
"The new Aveir pacing system holds the potential to help us advance treatment for people who need a pacemaker but where physicians are concerned about more invasive surgery or in circumstances where we believe a person's therapy needs may change in the future and where retrievability will be a critical device feature," Reddy said.
The Food & Drug Administration is evaluating Aveir.
Because life can move prestissimo (very quickly) and the beat can change, having the ability to bring in a new conductor will help keep the orchestration of your life.
And for Aveir, no baton required.
Bravo!
For the latest on Abbott’s life-changing technology, get updates directly in your inbox.