In a development that substantially expands the availability of the FreeStyle Libre and FreeStyle Libre 14 day system app (FreeStyle LibreLink) to more people, the U.S. Food and Drug Administration (FDA) has approved use for Android users. Since the FreeStyle LibreLink Android version recently became available it has soared to one of the top 10 medical apps on the Google Play store.1
Now, combined with the previously approved iPhone iOS apps, many people using a compatible smartphone2 can manage their diabetes care. This expanded access is another important step forward as Abbott continues to advance its digital ecosystem for the FreeStyle Libre system and FreeStyle Libre 14 day system.
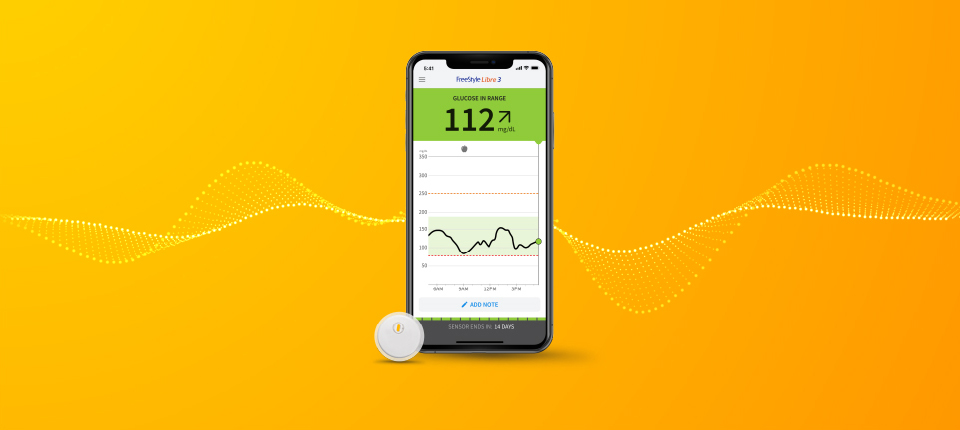
Many smartphone users living with diabetes will be able to monitor their glucose levels directly through FreeStyle LibreLink,3 obtaining current glucose in seconds — including their eight-hour glucose history as well as tracking changes to their glucose levels. The app eliminates the need for a separate hand-held FreeStyle Libre or FreeStyle Libre 14 day reader,4 which makes checking your glucose even more convenient, having one less thing to carry around.
Availability of the FreeStyle LibreLink app enhances the value of the FreeStyle Libre and the FreeStyle Libre 14 day systems, which have proven to ultimately help users achieve better health outcomes,5 while eliminating the need for painful fingersticks.6
These systems are designed to improve the lives of people living with diabetes, building on Abbott's commitment that helps more than 1.5 million FreeStyle Libre users in 46 countries, every day.7
The FreeStyle LibreLink app is compatible with NFC-enabled smartphones running Android OS 5.0 or higher and iPhone 7 or later running iOS 11 or later.
FreeStyle, Libre, and related brand marks are trademarks of Abbott Diabetes Care Inc. in various jurisdictions. Other trademarks are the property of their respective owners.
Android is a trademark of Google LLC.
Google Play are trademarks of Google LLC.
References
1Data on file. Abbott Diabetes Care. Ranked #9 as of June 12, 2019.
2The FreeStyle LibreLink app is compatible with NFC-enabled smartphones running Android OS 5.0 or higher and iPhone 7 or later running iOS 11 or later.
3Use of the FreeStyle LibreLink app requires registration with LibreView, a service provided by Abbott and Newyu, Inc.
4The FreeStyle LibreLink app and the FreeStyle Libre and FreeStyle Libre 14 Day reader have similar but not identical features. Fingersticks are required for treatment decisions when you see Check Blood Glucose symbol, when symptoms do not match system readings, when you suspect readings may be inaccurate, or when you experience symptoms that may be due to high or low blood glucose. When using FreeStyle LibreLink app, access to a blood glucose monitoring system is required as the app does not provide one.
5Data on file. Abbott Diabetes Care.”
6Fingersticks are required for treatment decisions when you see Check Blood Glucose symbol, when symptoms do not match system readings, when you suspect readings may be inaccurate, or when you experience symptoms that may be due to high or low blood glucose.
7Data on file. Abbott Diabetes Care.




FOLLOW ABBOTT