New Data Show TriClip’s Treatment Potential
Study finds Abbott's first-of-its-kind TriClip, an investigational device in the U.S., improves quality of life compared to medical therapy alone for tricuspid regurgitation, a heart condition with historically few treatment options. ,
Mar. 30, 2023
- Copy Link
- Share on X
- Share on Facebook
- Share on Linkedin
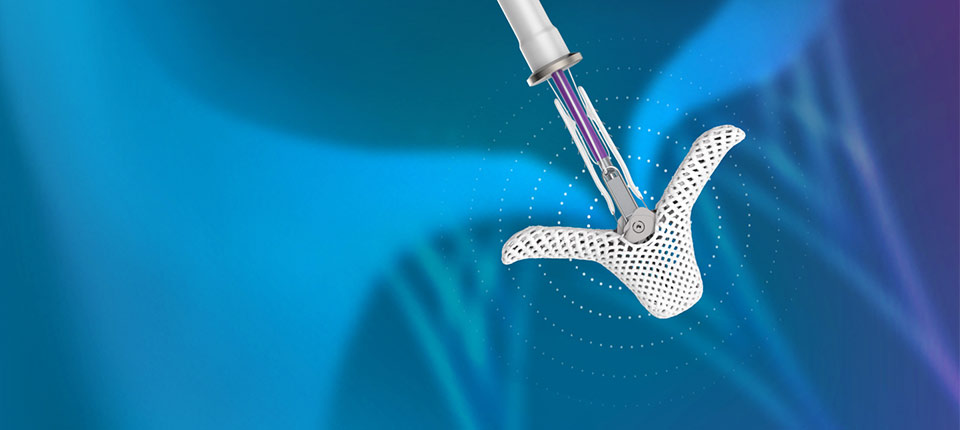
Left or right? The difference matters in turn lanes, penmanship and heart conditions. One of the innovations in Abbott's structural heart portfolio focuses on treating a leaky tricuspid valve on the right side of the heart.
Most cardiac interventions target the left side of the organ, meaning that someone with tricuspid regurgitation (TR), a condition caused by a defective tricuspid valve (which controls the flow of blood from the right atrium to the right ventricle), has very few treatment options. When left unaddressed, TR can lead to atrial fibrillation, heart failure and, possibly, death.
Abbott's TriClip transcatheter edge-to-edge repair system, which first received CE Mark approval in April 2020, is a first-of-its-kind minimally invasive device that reduces the backflow of blood caused by TR and allows physicians to tailor the repair of the valve to each patient’s unique anatomy.
Promising Study Results on TriClip
Findings from the TRILUMINATE Pivotal trial, which began in 2019, were shared in early March this year at the American College of Cardiology 72nd Annual Scientific Session together with World Congress of Cardiology. At a high level, the results demonstrate TriClip was superior to medical therapy with significant improvements in quality of life and TR. The findings show TriClip demonstrated safety and met the primary endpoint. Mortality or tricuspid valve surgery and heart failure hospitalizations did not appear different between the groups at one year.
TriClip is approved for use in more than 50 countries, including Europe and Canada, and is currently an investigational device in the United States.
"Tricuspid regurgitation is considered the most undertreated valve issue, and the TRILUMINATE Pivotal data show how TriClip is helping to restore health and improve the quality of life for people with structural heart disease," said Michael Dale, senior vice president of Abbott's structural heart business.
For the latest on Abbott’s life-changing technology, get updates directly in your inbox.